Our values also stand for our products and applications
Sterilizer applications
Steam-air Mixture Process
The steam-air mixture process is optimised for all liquids in closed and open containers as well as solids and is based on many years of experience in the development and optimisation of sterilisers for the highest demands. Typical application is the sterilisation of syringes, ampoules, stoppers, pouches, reagents.
The process was developed to sterilise non-vacuum-tight pouches and syringes. If these were sterilised with the common saturated steam method, what would happen can be seen in the video.
Sterilisation Goods in Foil Bags
Many products to be sterilised are wrapped in foil pouches. With the Belimed Life Science sterilisation processes, however, these products can also be reliably sterilised.
What happens to the pouch in the process is shown in the video. The process in the video is shown in fast motion.
Sterilisation of Syringes
Syringes are a particular challenge in sterilisation. Since there is still a small amount of air in the syringes, there is a risk that the stopper in the syringe will move. To prevent this from happening, syringes are sterilised using the Belimed Life Science steam-air mixture process with support pressure, otherwise what is shown in the video could happen.
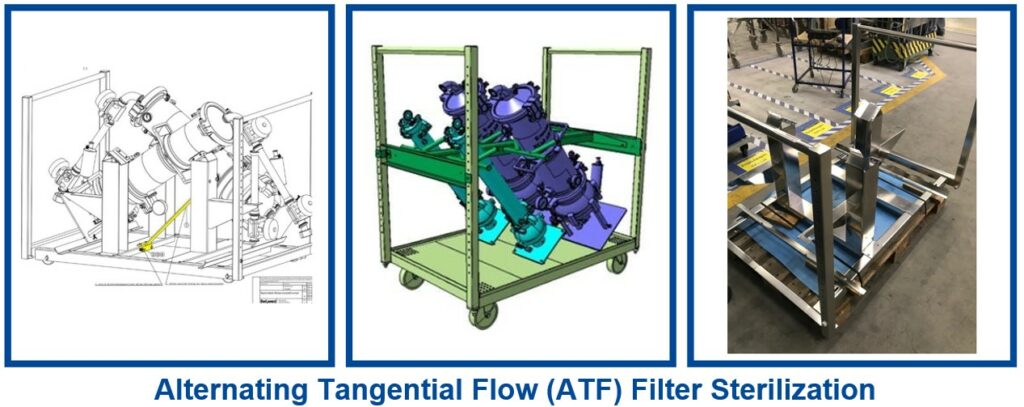
ATF Filter Sterilisation
There is a growing trend to sterilize ATF (Alternating Tangential Flow) filters and their associated tubing. The challenge is to find the right sterilization processes and the optimal sterilization position. Belimed Life Science has developed proven sterilization programs that provide reliable and repeatable sterilization processes. This ATF filter sterilization process can be optimized in our widely accepted Belimed Life Science BST and PST sterilizers. In addition, Belimed Life Science has developed special racks that accommodate as many ATF filters as possible to fit into a dedicated sterilizer. The selected angle has been calculated to allow the best sterilisation results and optimal use of space.
Cleaning System Applications

Most modern GMP-compliant container cleaning in pharmaceutical production
Depending on the process requirements and the type of items to be washed, various methods of loading and unloading the wash chamber are possible. Individual customer requirements can be taken into account. For example:
- Manual loading and unloading system.
- Automatic loading and unloading system.
In the development of the PH 880.2, the focus is on a design that takes customer requirements into account and at the same time complies with the latest FDA, GMP and GAMP guidelines.

Cleaning with rotating wash ware carriers
If many small and complex components have to be cleaned, Belimed Life Science has developed a solution with a rotating wash ware carrier. The wash ware carrier rotates like a Ferris wheel and ensures that each wash ware receives the same cleaning performance. This ensures a high degree of process repeatability.
The PH 860.2 cleaning system even offers space for 2 wash ware carriers of this type to increase throughput.

Cleaning system with intervention function for safe dismantling of the items to be washed
If a heavily contaminated wash item has to be disassembled before the cleaning process, the solution developed by Belimed Life Science with an intervention function is the ideal solution. The focus is on protecting the environment and the operator. To ensure this, the wash item is first positioned in the cleaning system and the doors are closed. Two gloves integrated into the system wall can then be used to safely dismantle certain components on the items to be washed. There is also an extra storage place for the dismantled parts so that they are also cleaned during the cleaning process.
After cleaning, the components can be reassembled in exactly the same way or the assembly can take place outside the cleaning system.